- Blog
- Bose speaker enclosure design
- Dragon ball z kai season 5 episode -1 eng dub
- Bruce springsteen easy guitar songbook
- Grand theft auto iv download free
- Docker run as root in container
- Truth or dare sexual videos
- Naruto shippuden ultimate ninja 5 ps2 save file
- Skip bo rules japanese
- 3d xps peak ratio
- Harvey danger radio silence
- Digidesign digi 003 rack
- Shadowsocks client linux

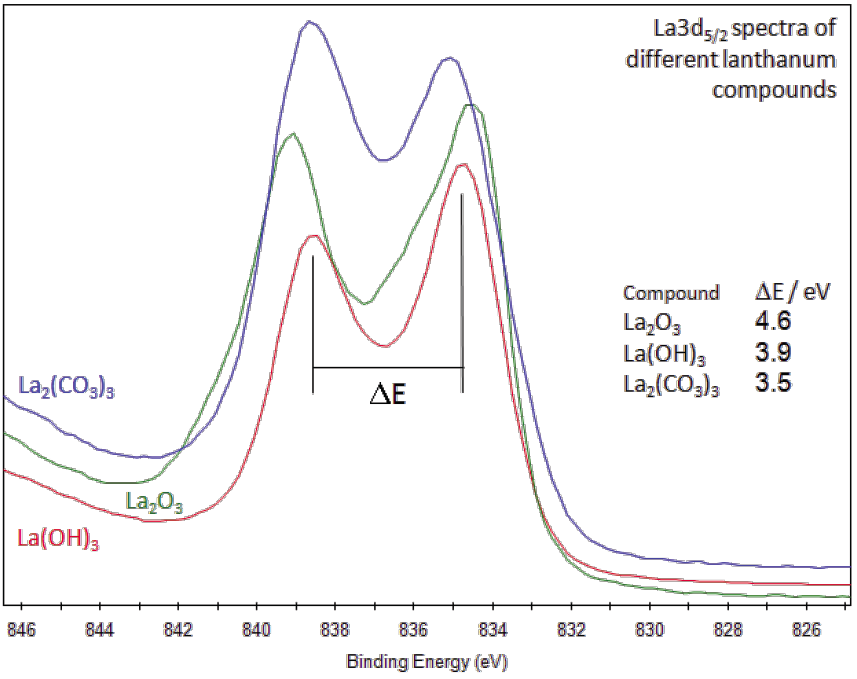
The XPS analysis were done using Al Ka (hv =1486.4 eV) radiation.ġ00 AvA NAN Aud AG MEAN FREE PATH (Å) Aua Ago Aga Au Aud со Age. Use the Electron Mean free path and cross sections data given in the attached file. Calculate the atomic concentration ratio of the Si/Ta and N/Ta. The peak area of the "Sizp" peak is 1000 and the peak area of the “Nis” peak is 400. The XPS peak area under the "Tai" peak is 5000 counts. He has authored 800 papers for a Web-of-Science h-index of 72, and holds 25 patents from active collaboration with industry for wear-resistant cutting tools, low-friction coatings for components, thin film neutron detectors, and diffusion barriers in microelectronics.Transcribed image text: 4. The intensity of the N+ peak decreases as the material loses its conductivity, hence the ratio of the experimentally derived N+ /C atomic ratio correlates. He has served as Director for several Centers of Excellence in Materials Research. This arises from spin-orbit coupling effects in the final state. Recognitions include the European ERC Advanced Grant, a Wallenberg Scholar Grant, the Akzo Nobel Sweden Science Award, and Fellow of the American Vacuum Society. The 3d photoemission is in fact split between two peaks, one at 334.9 eV BE and the other at 340.2 eV BE, with an intensity ratio of 3:2. He has procured and managed the electron microscopy laboratory at Linköping, including the FEI Analytical Monochromated Double-aberration-corrected 60-300 kV Titan 3 high-resolution instrument and its dedicated vibration-free building. His research interest is in materials science and nanotechnology of functional thin film materials. He made a sabbatical at University of Illinois at Urbana-Champaign during 2004-2006. He received his PhD 1988 and became Professor/Head of Division in Thin Film Physics at IFM, Linköping University in 1998 after being a visiting scientist to Northwestern University, USA. He is an elected member of the Royal Swedish Academy of Science (KVA) and Engineering Sciences (IVA), as well as Fellow of the American Vacuum Society and the Forschungszentrum Dresden-Rossendorf. He is leave of absence with time for research from Linköping University, Sweden. He is Chief Executive Officer of The Swedish Foundation for Strategic Research (SSF), since 2013. Lars Hultman, Born 1960 in Linköping, Sweden. Our intention is to promote awareness within a growing XPS community as to the problems reported over the last six decades and present a guide with best practice for using the C 1s BE referencing method. has 5 unpaired electrons in the 3d shell as shown below. Owing to the huge volume of XPS literature produced each year, the consequences of improper referencing are dramatic. Auger peaks produced by x-rays (transitions from L to K shell: O KLL or. The purpose of this review is to critically evaluate the status quo of XPS with a historical perspective, provide the technique’s operating principles, resolve myths associated with C 1s referencing, and offer a comprehensive account of recent findings. Alarmingly, as revealed by our survey of recent XPS literature, the calibration procedure based on the C 1s peak of adventitious carbon is highly arbitrary, which results in incorrect spectral interpretation, contradictory results, and generates a large spread in reported BE values for elements even present in the same chemical state. Despite accumulating criticism, pointing to the unknown origin and composition of the adventitious carbon, this is by far the most commonly used method today for all types of samples, not necessarily electrically insulating. One approach, proposed in the early days of XPS, employs the C 1s spectra of an adventitious carbon layer, which is present on all surfaces exposed to air. The accuracy of chemical-state determination relies, however, on a trustworthy calibration of the binding energy (BE) scale, which is a nontrivial task due to the lack of an internal BE reference. With more than 9000 papers published annually, X-ray photoelectron spectroscopy (XPS) is an indispensable technique in modern surface and materials science for the determination of chemical bonding.
- Blog
- Bose speaker enclosure design
- Dragon ball z kai season 5 episode -1 eng dub
- Bruce springsteen easy guitar songbook
- Grand theft auto iv download free
- Docker run as root in container
- Truth or dare sexual videos
- Naruto shippuden ultimate ninja 5 ps2 save file
- Skip bo rules japanese
- 3d xps peak ratio
- Harvey danger radio silence
- Digidesign digi 003 rack
- Shadowsocks client linux
